 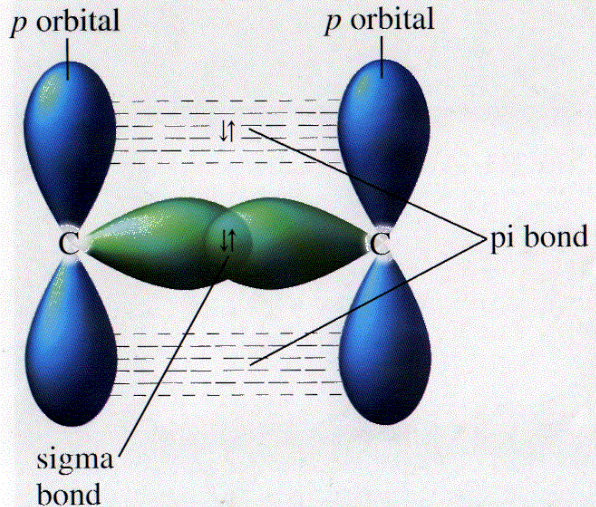
The unhybridized 2 p atomic orbitals overlap with each other side by side, and they form a pi bond ( bond) that has a single nodal plane that coincides with the molecular plane of the. Since the head-to-head overlap occurs along the internuclear region, it has a larger overlapping area.Īlso, once the bond formation completes, the bond undergoes symmetric rotation along the internuclear axis.Ĭommon orbital overlaps forming a sigma type of covalent bond are- s+s, p z+p z, s+p z, and d z 2+d z 2 (where z is defined as the axis of the bond or the internuclear axis). There is a single sigma bond between the two carbon atoms in ethene, and each carbon atom forms another two sigma bonds with adjacent hydrogen atoms. When two similar or dissimilar atomic orbitals approach along the central line or axis of the nucleus, known as the internuclear axis, it leads to a direct head-to-head overlap of the atomic orbitals creating the strongest, most efficient single covalent or the sigma bond.įor example, the head-on-head overlap of the two s-orbitals and two p-orbitals along the internuclear axis is shown below. Carbon atoms share these four electrons in four covalent bonds with other atoms to achieve a full outer shell configuration These electrons are found in orbitals within the respective atoms When forming a covalent bond, the orbitals overlap in such a way to form two types of bonds. The sigma bonds are usually mentioned in valence bond theory to visualize the bond formation between atoms to form polyatomic molecules like H 2, CH 4, etc., by overlapping their atomic orbitals. 
The single covalent bond is referred to as a sigma bond, denoted by the symbol σ. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
